Refrigerants are called material cools them to be cooled by taking heat from material in general. Refrigerants surrounding substances are cooled directly or indirectly.
Coolers are sought after features for fluid;
– Must be positive evaporating pressure. It should be slightly above the ambient pressure of the vapor pressure to prevent business disruptions not meyden to solidify in the cold part of the water vapor.
– Must be low condensation pressure. High pressure compressor, condenser, such as pipeline installation should be.
– Latent heat of vaporization must be high. Latent heat of the higher rate of gas that will be used in the system fluid.
– Chemically should be active, do not affect the installation material to be corrosive, should not change the property of lubricating oil.
– Should not flammable explosive and poisonous.
– Leakage of enabling the feature should be easy to identify. (Odor, color)
– It should be cheap.
– Thermal conductivity must be high.
– D should be electric.
– Low freezing temperature should be.
– It must be high critical temperature.
– Specific volume should be smaller.
– The viscosity should be low.
– At atmospheric pressure, it must be able to evaporate at a low temperature.
– Should not be high condensing pressure. Condensation temperature should be high.
– Coolant should not affect the system’s pipes.
– Flowing chemical structure should be stable in the System.
must not change the properties of the lubricating oil in -Kompresör.
– It should not be toxic.
– Small capacity should be suitable for the use of a compressor.
First Class Refrigerants
Many species of the most diverse operating conditions of refrigerant vardır.bun and in order to ensure compliance with the requirements and were specifically developed. Some refrigerants are mixed with a chemical structure and are usually symbolized by refrigerant classification numbers beginning with the letter R. Some refrigerants commonly used classification numbers and some physical properties are given in the following table.
| Gases | 760 mm kaynamaºc | Critical temperature ° C | Critical pressure (atm) | Freezing point ºC |
| Oxygen | -183 | -18.5 | 49.7 | -218 |
| Nitrogen | -195.65 | -146.74 | 33.54 | -200.95 |
| Hydrogen | -252.74 | -240 | 12.8 | -259 |
| Helium | -270 | -268 | 2.26 | -271 |
| Carbon dioxide | +31 | 73 |
Loss Certain physical properties of refrigerants

– R11: (ccl3f), low pressure (0 ° C 0.40 bar) is a cooler. Predominantly 350 kW – 10,000 kW cooling capacity range in which the centrifugal water chiller unit (chiller) is used. Full R11 is estimated that 60,000 units used in water cooling unit in the world. Production was stopped due to ozone depletion.Non-flammable and odorless.
– R12: So far, with the purpose of cooling was one of the most widely used refrigerant. However, the use now because of their negative impact on the ozone layer is a refrigerant which is prohibited.Besides, toxic, explosive and flammable completely safe due to lack substance.
– R123 (cf3chcl2) is used and the most appropriate alternative refrigerant R11 in centrifugal chiller unit. R11’Evaporat is more power to influence non-metallic materials. Therefore, R123 ‘ evaporator switch should be replaced all the rubber-based material. Based R11’Evaporat has lower energy efficiency. In its operating environment require additional measures because of the poisonous property. Exposure to maximum dose is 10 ppm for 8 hours.
– R134a (CF2CH2F), the thermodynamic and physical properties of R12 closest cooler. The ozone depletion coefficient is still the most suitable refrigerant for the 0 and other features. Vehicle cooling and domestic alternative is the most suitable for the cooler. R134a, is not compatible with mineral oil or poliolalkalinglikolbazl should be used with polyolester oil.
– R401 to R22, R124 and R152a consisting of (by weight were 52% / 33/15 rate) and a zeotropic mixture of acceptable alternative to R12. That HCFCs may be used until 2030 contained not a final alternative. This cooler has been introduced by name by DuPont SUVA MP39.
– R402A: R22, R125 and R290 consisting of (by weight, respectively 38% / 60/2 ratio) and a zeotropic mixture of acceptable alternative for R502. That HCFCs may be used until 2030 contained not a final alternative. This cooler has been introduced by DuPont SUVA HP80 by name.
– R404 R32 R125 and comprising (weight% 50/50 ratio) and a near azeotropic mixture accepted alternative to R22. Theoretical thermodynamic properties are not as good as R22. However, heat transfer capability is very good. R22 – R410 transformation is necessary to redesign the system. If this change is made in the efficiency of the system it is better than 5% compared to the R22. The high greenhouse effect is the biggest disadvantage.
– R407C: 407C, R32, R125 and consisting of R134a (wt% respectively 20/40/40, 10% / 70/20, and 23% / 25/52 ratios) and are a zeotropic mixture acceptable alternative to R502.
– R410 R32 R125 and comprising (weight% 50/50 ratio) and a near azeotropic mixture accepted alternative to R22. Theoretical thermodynamic properties are not as good as R22. However, heat transfer capability is very good. R22 – R410 transformation is necessary to redesign the system. If this change is made in the efficiency of the system it is better than 5% compared to the R22. The high greenhouse effect is the biggest disadvantage.
– R500: an azeotropic mixture of R12 and R152a. Mixing ratio by weight of 73.9% R12, R152a is 26.2%. Low rates of R12 is used as an alternative. better COP by R12 and 10 – 15% has a higher volumetric cooling capacity.
– R502: an azeotropic mixture of R22 and R115. Mixing ratio by weight of 48.8% R22, 51.2% R115’T is. Most used are refrigerated transport and commercial refrigeration field. at low temperatures it has a high volumetric cooling capacity. -20, Are higher than R22 in a range between -40 ° C and 1% to 7%. The COP of R22, depending on the operating conditions of 5% – 15 higher.
– R507: R125 and consisting of R134a (wt% 50/50 ratio) is an acceptable alternative for R502. This cooler has been released by the name Genetron AZ50 ALLIED SIGNAL.
– R717 (NH3 = ammonia): Ammonia is malodorous and colorless gas consisting of nitrogen and hydrogen. Chemical formula NH3. fertilizers containing nitrogen, pharmaceuticals, paints, is the first step in the synthesis of perfumes. Although the OH- ions comprises shows weak base functionality.hidden in the gasification temperature is too high when used as a coolant in industrial plants.Molecular weight 17.0304 g / mol, is -33.34oc boiling point at 1 atmospheric pressure (239.81 K).Molecular shape is a triangular pyramid. highly soluble in water is the polar molecules. In cases where more importance to the toxic effects of large-capacity plant, cold warehouse, the production of ice, the ice-skating rink and is widely used in packaging frozen applications.
– R13: (cclf3), low temperature refrigerant used between -70 0C to -45 0C. Few are used in industrial cooling installations.
– R13B1 (cbrf3) -45 0C is used in industrial refrigeration in the range of -700C. Because of the high ozone depleting capacity of production and consumption under the Montreal Protocol it has been completely stopped.
– R114 (cclf2) is non-combustible and toxic refrigerant. 80 0C – 120 0C is used in industrial heat pump.
– R143 A: (CF3CH3) for long periods R502 and R22 are acceptable alternatives. The use of ammonia is used in low temperature applications is not suitable.
– R125 (CF3CHF2), R502 and R22 for the long term has been accepted as an alternative. It is not appropriate use of ammonia as R143 is designed for low temperatures.
– R152 to: do not cause damage to the ozone and the greenhouse effect, which is very low (up to 2% of R12) to R152 (C2H4F2), was adopted as an alternative to R12 and R500 heat pumps.
Other Refrigerants
Recently, a type developed azeotrope refrigerant (mixed) refrigerants are mixed in a certain ratio of two refrigerants were in a single refrigerant such as mad available refrigerant. Evaporation and condensation are no changes in the structure during. In systems where the compressor is directly connected to the electric motor, the power source frequency changes the cooling capacity varies.This change can be avoided if azeotrope refrigerant used.
Salt solutions donors at lower temperature from the freezing point of pure water. chilled saline solution with direct coolant is transported by pipes to the cold storage or ice-making system. Taking heat from the material to be stored in cold and cools them. It is most commonly used with calcium chloride brine solution.
| Refrigerants | Composition (by weight) |
| R401 to | R22 + 33% + 15% 52% R124 to R152 |
| R402A | R22 + 60% + 2% 38% R125 R290 |
| R404A | R125, R134a 44% + 4% + 52% to R143 |
| R407 to | R125 + R32 + 40% 20% 40% R134a |
| R407B | R32 + R125 + 20% 10% 70% R134a |
| R407C | R125 + R32 + 23% 25% 52% R134a |
| R410A | R125, R32 + 50% 50% |
| R500 | R12 to R152 73.8% + 26.2% |
| R502 | R115 + R22 51.2% 48.8% |
| R507 | R143 to R125 + 50% 50% |

Table: refrigerant tubes color chart
Refrigerant Leakage Control Methods
– Bubble
All the gas in the leak may be used.
– Torch Lamb the (halidelamp)
All fluorocarbon refrigerant used for. Leakage would open flame is blue-green.
– Electronic leak detector
All fluorocarbon refrigerant used for. Balance has automatic or preset types. (Note: Ammonia leaks some discoloration of litmus paper-based methods, such as white smoke and sulfur wax is used to remove the smoke.)

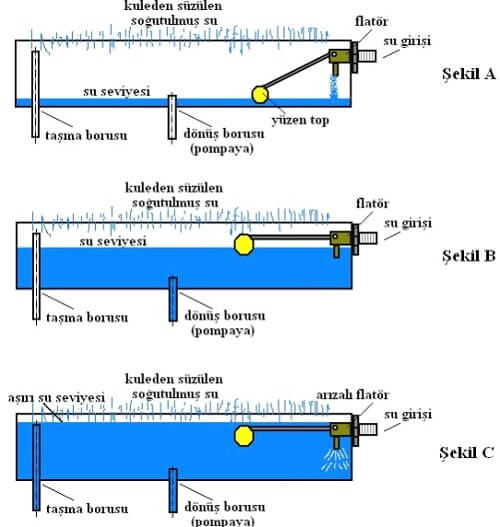


The article has actually peaks my interest. Thanks.